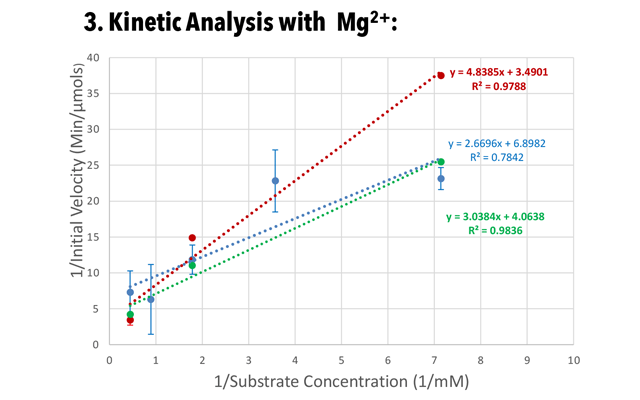
Calf intestinal alkaline phosphatase (CIP) is an enzyme that catalyzes the removal of phosphate groups from the 5′ of nucleic acids. This prevents the re-ligation of linearized DNA in the creation of cloning vectors and helps increase the yield of usable vectors. (Promega, 2018). The enzyme’s activity is important in subcloning and prepping for DNA sequencing and SNP analysis, so the aim of this project is to study the effect of the addition of the activator Mg2+ on the kinetics of CIP and determine its efficiency as an activator. We hypothesis that AP activity increases with the addition of Mg2+, and that we will see an increase in Vmax, which is the reaction rate when the enzyme is fully saturated by substrate, and a decrease in Km, which is an indicator of affinity and equal to substrate concentration at half maximum velocity.